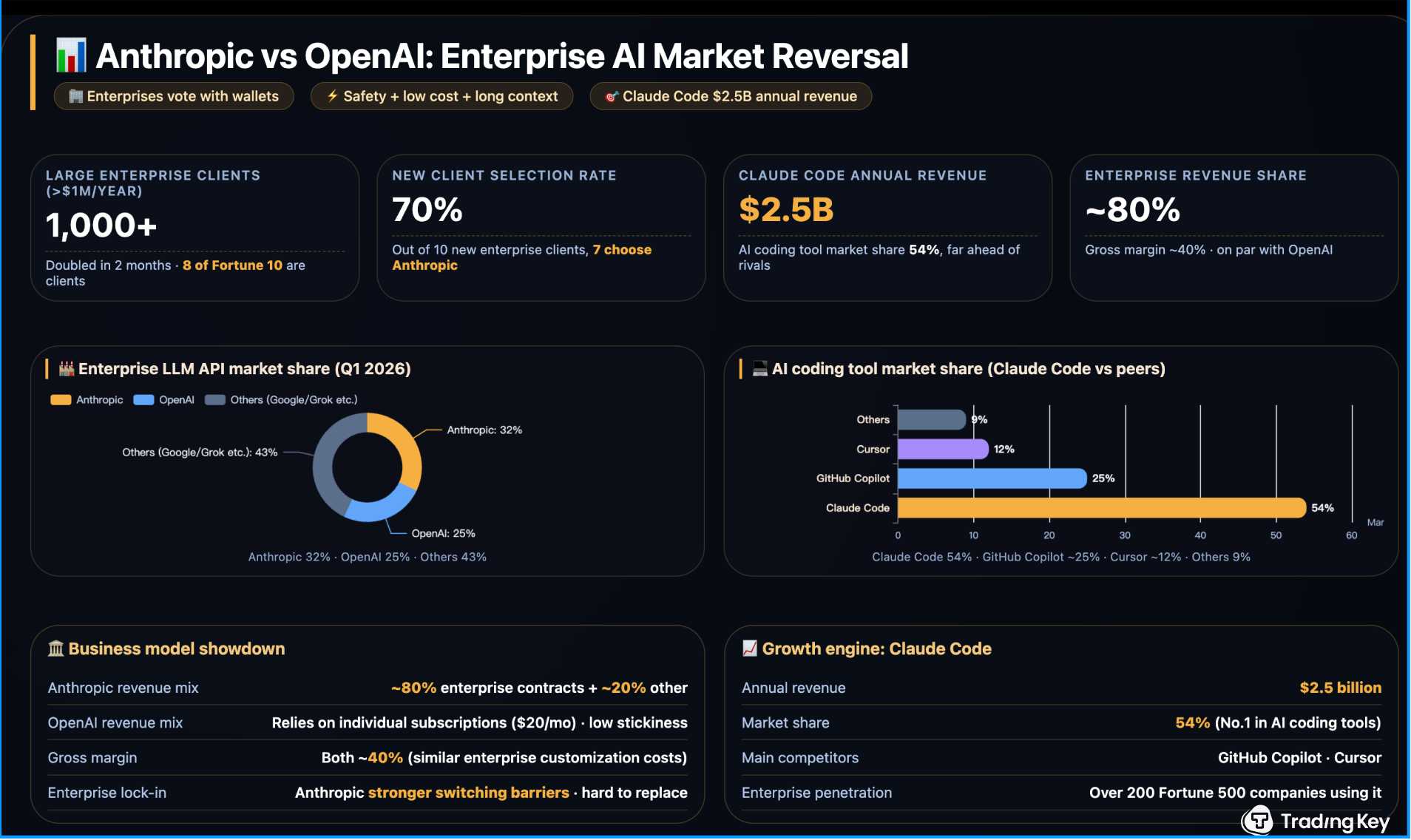
Adagio Medical rises after FDA clears expanded testing of heart device
Shares of medical device maker Adagio Medical ADGM.O rise 11% to $1.31 premarket
Co says the FDA approved a clinical sub-study of its experimental vCLAS Ultra-Low Temperature Ablation system
The device targets sustained monomorphic ventricular tachycardia, a dangerous heart rhythm that can lead to sudden cardiac arrest - ADGM
Co says the system uses extreme cold to destroy faulty heart tissue and is designed to shorten procedure times and improve handling for doctors
The sub-study will enroll up to 55 patients with ischemic and non-ischemic heart disease, part of Adagio’s ongoing pivotal U.S. trial - ADGM
Co says the technology is already approved in Europe and is being evaluated in a larger U.S. study
As of last close, stock up ~13% YTD
Recommended Articles