Sanofi SA Stock (SNY) Moved Up by 3.33% on Mar 25: Key Drivers Unveiled
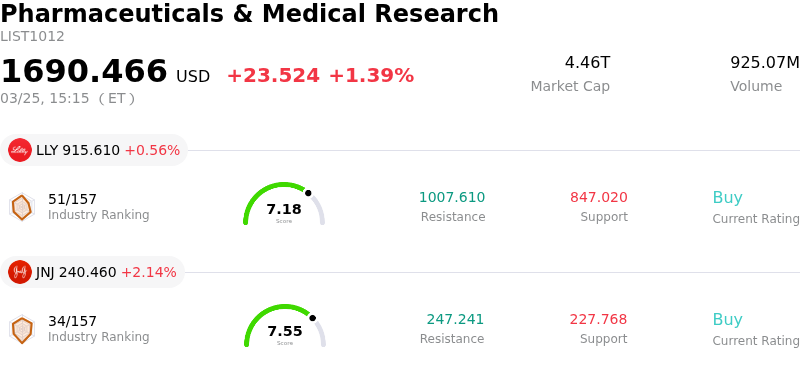
Sanofi SA (SNY) moved up by 3.33%. The Pharmaceuticals & Medical Research sector is up by 1.39%. The company outperformed the industry. Top 3 stocks by turnover in the sector: Terns Pharmaceuticals Inc (TERN) up 5.85%; Eli Lilly and Co (LLY) up 0.56%; Johnson & Johnson (JNJ) up 2.14%.
What is driving Sanofi SA (SNY)’s stock price up today?
Sanofi (SNY) experienced upward price movement today, driven by a confluence of positive developments related to its pharmaceutical pipeline and regulatory landscape.
A significant catalyst was the announcement on March 24, 2026, that the US Food and Drug Administration (FDA) granted Breakthrough Therapy designation to venglustat, an investigational oral therapy for neurological manifestations of type 3 Gaucher disease. This designation is based on compelling Phase 3 clinical trial results, which demonstrated statistically significant neurological improvement. Breakthrough Therapy designation is intended to accelerate the development and review process for promising new medicines addressing serious or life-threatening conditions, signaling a potentially faster path to market for this rare disease treatment.
Further bolstering investor confidence, Sanofi and its partner Regeneron received approval from Japan's Ministry of Health, Labour and Welfare for Dupixent (dupilumab). This approval allows Dupixent to be used for the treatment of adults with moderate-to-severe bullous pemphigoid, a debilitating skin condition. This expands the market reach for one of Sanofi's key growth drivers, following positive data from a pivotal Phase 2/3 study.
Additionally, on March 24, 2026, Sanofi announced a significant licensing agreement with Kali Therapeutics. This deal grants Sanofi exclusive global rights to KT501, a novel tri-specific antibody for B cell-mediated autoimmune diseases. The agreement includes substantial upfront and near-term payments, alongside potential future milestone payments, indicating a strategic expansion of Sanofi's immunology pipeline.
Pre-market sentiment on the trading day also saw a positive reaction among insulin manufacturers, including Sanofi, following reports that a bipartisan group of US senators reached a deal to cap insulin prices. While the full implications of such legislation are complex, the market initially perceived this as a factor contributing to positive movement for companies in this sector.
Overall, the combination of a significant FDA designation for a rare disease drug, expanded market approval for a blockbuster product, and a strategic pipeline acquisition appears to have fueled the positive investor sentiment for Sanofi today. These developments collectively highlight the company's progress in clinical development and market expansion. Analysts maintain a "Buy" consensus rating for Sanofi, anticipating further upside.
Technical Analysis of Sanofi SA (SNY)
Technically, Sanofi SA (SNY) shows a MACD (12,26,9) value of [-0.76], indicating a neutral signal. The RSI at 47.74 suggests neutral condition and the Williams %R at -17.27 suggests oversold condition. Please monitor closely.
Fundamental Analysis of Sanofi SA (SNY)
Sanofi SA (SNY) is in the Pharmaceuticals & Medical Research industry. Its latest annual revenue is $52.67B, ranking 10 in the industry. The net profit is $8.81B, ranking 7 in the industry. Company Profile
Over the past month, multiple analysts have rated the company as Buy, with an average price target of $59.19, a high of $69.14, and a low of $53.00.
More details about Sanofi SA (SNY)
Company Specific Risks:
- Financial vulnerability from significant reliance on Dupixent, which faces a patent cliff beginning in 2030, posing a long-term threat to a product that accounts for approximately 30% of Sanofi's total sales.
- Sustained underperformance in share price, with the stock experiencing a 25.8% decline over the last year as of mid-March 2026 and trading near its lowest levels since March 2021, reflecting ongoing investor concerns and market pressure.
- Lingering investor apprehension regarding drug pipeline execution, stemming from previous FDA delays and failed Phase 3 trials for key assets like tolebrutinib in late 2025, which continues to raise questions about R&D productivity and future sales estimates.
- Strategic uncertainty introduced by recent changes in top leadership during February 2026, potentially impacting the company's ability to address ongoing drug pipeline challenges and declining vaccine sales effectively.