Plus Therapeutics rises as FDA grants orphan drug tag for pediatric brain cancer treatment
Shares of biotech firm Plus Therapeutics PSTV.O rise 15% to $3.36 premarket
Co says the FDA granted orphan drug designation to experimental drug reyobiq for pediatric malignant gliomas, a rare and aggressive childhood brain cancer, which is hard to treat with surgery or chemotherapy alone
Co says reyobiq delivers targeted radiation directly into tumors to limit damage to healthy brain tissue
The FDA has cleared the drug for testing in children; studies are also ongoing in other brain cancers and cancer spread to brain fluid - PSTV
FDA grants orphan drug status to promote drug development for rare diseases affecting fewer than 200,000 people in the U.S.
As of last close, stock down ~77% YTD
Disclaimer: The information provided on this website is for educational and informational purposes only and should not be considered financial or investment advice.
Recommended Articles
Featured Tools
Top News
SpaceX IPO Guide: Can You Buy SpaceX IPO Shares on Robinhood? How Can Investors in Asia and Europe Buy SpaceX IPO Shares?

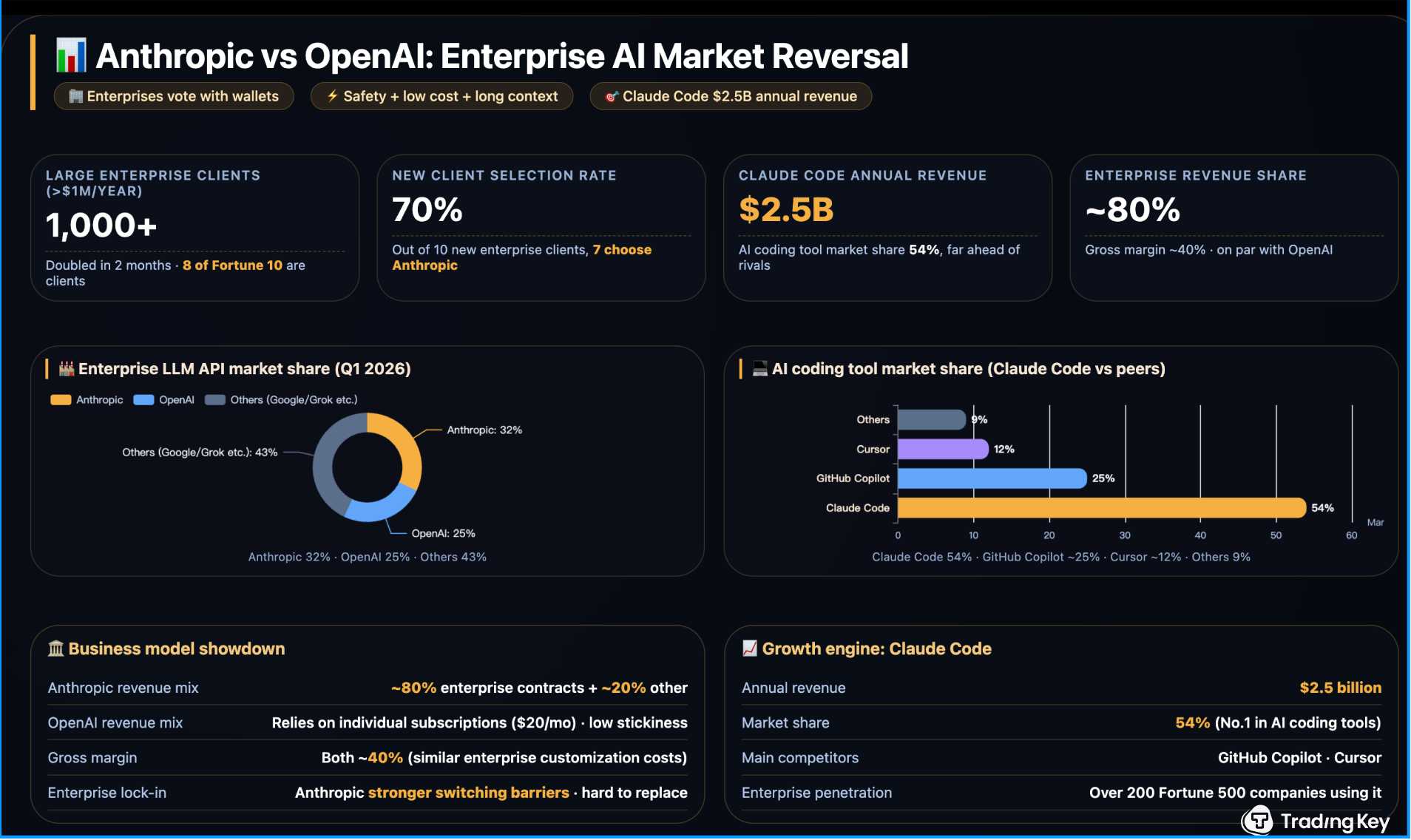
Anthropic Revenue Surpasses OpenAI for First Time, IPO as Early as October
Meta Is About to Launch a Consumer-Facing AI Model, and OpenAI Is About to Have Its IPO, Making Now a Good Time to Buy Meta Stock

IonQ vs. Rigetti Computing: Which Quantum Computing Stock Has the Edge Now?

Amazon Stock: 4 Pillars Supporting a Buy Thesis in a Cautious Market

Tradingkey







