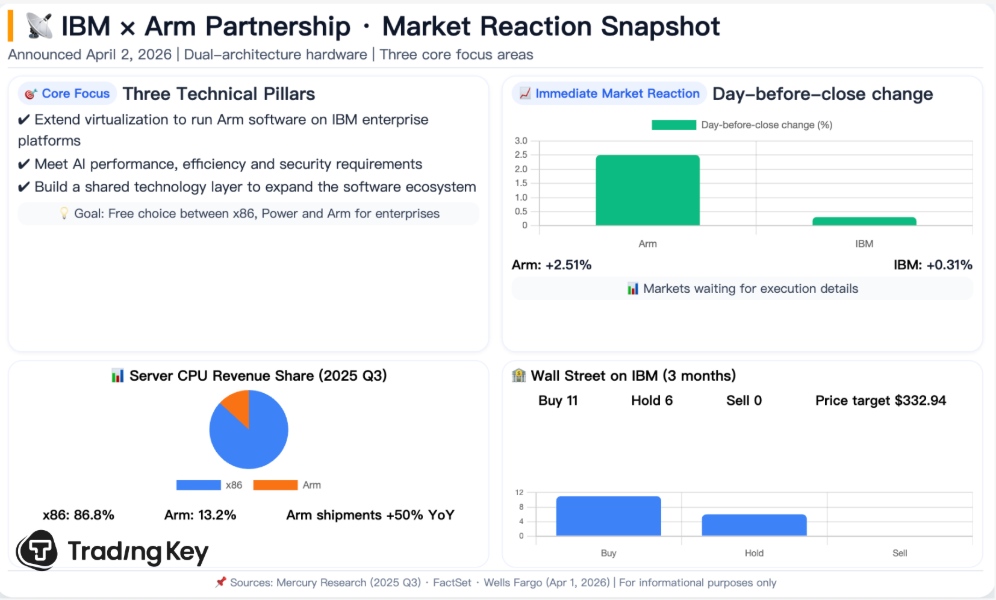
Ultragenyx rises after FDA accepts marketing application for rare disease therapy
Shares of drugmaker Ultragenyx Pharmaceutical RARE.O rise 1.96% to $21.84
The firm says the U.S. Food and Drug Administration has accepted to review its resubmitted application seeking accelerated approval for UX111, its gene therapy for a rare genetic disorder
Says updated long-term data in the filing include up to eight years of follow-up and show continued benefit across clinical measures and biomarkers, with an acceptable safety profile
Says if approved, UX111 would be the first approved treatment for Sanfilippo syndrome Type A, a rare disease in young children that causes severe brain damage and early death
UX111 is a one-time gene therapy designed to treat the underlying enzyme deficiency that drives the disease - RARE
Says the FDA is expected to make a decision by September 19, 2026
RARE shares fell more than 45% in 2025