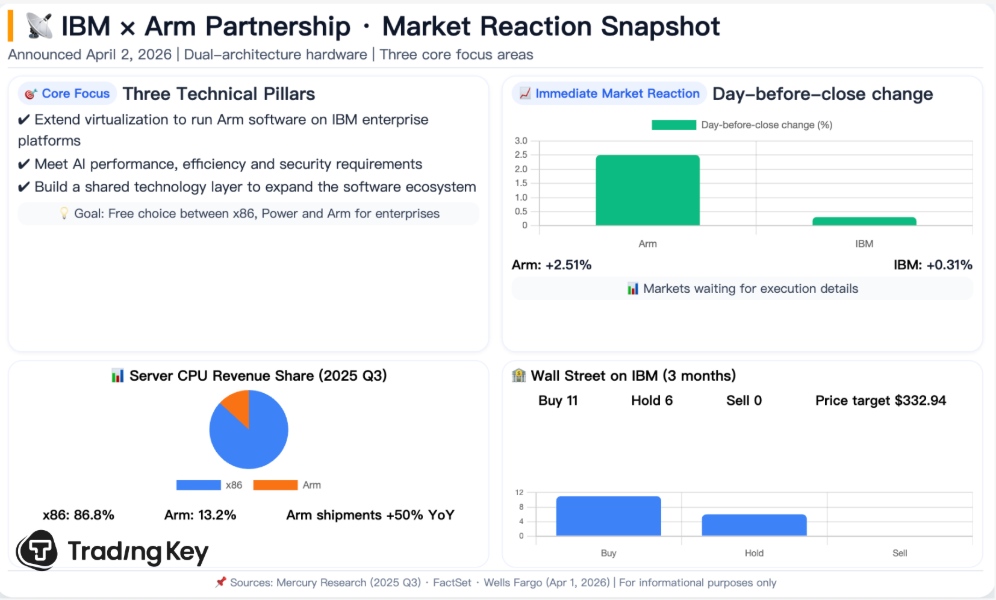
Cocrystal Pharma rises as FDA fast-tracks oral norovirus drug
Shares of drug developer Cocrystal Pharma COCP.O rise 9% to $1.10 premarket
Co says FDA grants fast-track status to its experimental oral drug CDI-988 for norovirus, a highly contagious stomach virus that causes vomiting and diarrhea; no approved treatments exist
CDI-988 is being tested in an early-stage study in up to 40 healthy adults to reduce norovirus symptoms and assess safety and how the drug moves through the body, co says
Says norovirus hits immunocompromised patients hardest, often causing long-term infections; CDI-988 is not yet approved
Fast-track status aims to speed development and review for drugs addressing serious diseases with unmet medical needs
As of last close, stock up ~4% YTD
Disclaimer: The information provided on this website is for educational and informational purposes only and should not be considered financial or investment advice.
Tradingkey
Recommended Articles
Tradingkey