Celcuity Stock Soars 112% as Its Breast Cancer Combo Hits Primary Endpoints, Teeing up FDA Filing
Celcuity stock soared more than 112% in premarket trading.
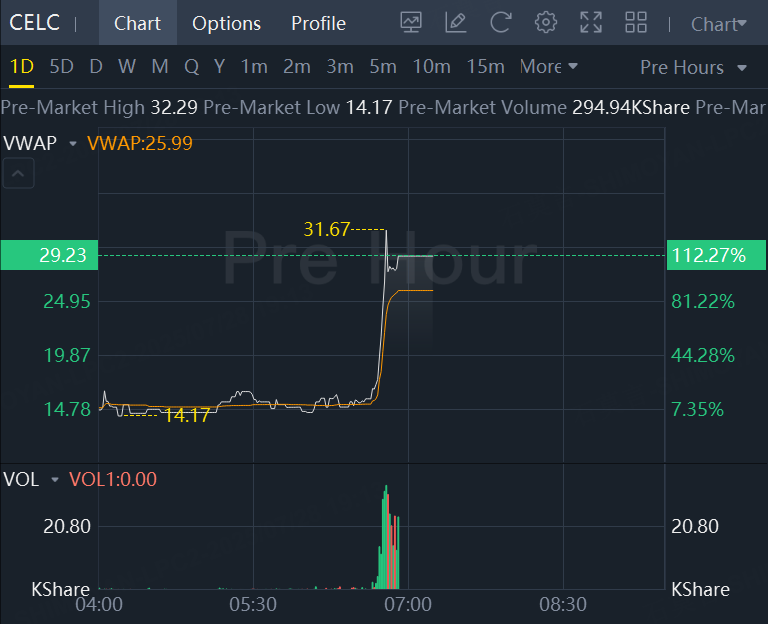
Celcuity announced breakthrough topline results from the PIK3CA wild-type cohort of its Phase 3 VIKTORIA-1 trial evaluating gedatolisib combinations for HR+/HER2- advanced breast cancer. The gedatolisib triplet therapy showed a 76% reduction in disease progression risk (HR=0.24) with 9.3 months median PFS vs. 2.0 months for fulvestrant alone. The gedatolisib doublet demonstrated a 67% reduction (HR=0.33) with 7.4 months median PFS.
These results represent unprecedented efficacy in HR+/HER2- advanced breast cancer treatment, with both combinations showing better safety profiles than previous trials. Treatment discontinuation rates were lower than observed in Phase 1b and other Phase 3 trials. Celcuity plans to submit a New Drug Application to the FDA in Q4 2025, with full data presentation scheduled for later this year.








