Bristol-Myers Squibb Canada Says New Subcutaneous Formulation Of Opdivo Approved In Canada
May 27 (Reuters) - Bristol-Myers Squibb Canada:
BRISTOL-MYERS SQUIBB CANADA: NEW SUBCUTANEOUS FORMULATION OF OPDIVO APPROVED IN CANADA FOR USE ACROSS ALL AUTHORIZED SOLID TUMOUR INDICATIONS
Disclaimer: The information provided on this website is for educational and informational purposes only and should not be considered financial or investment advice.
Recommended Articles
Featured Tools
Top News
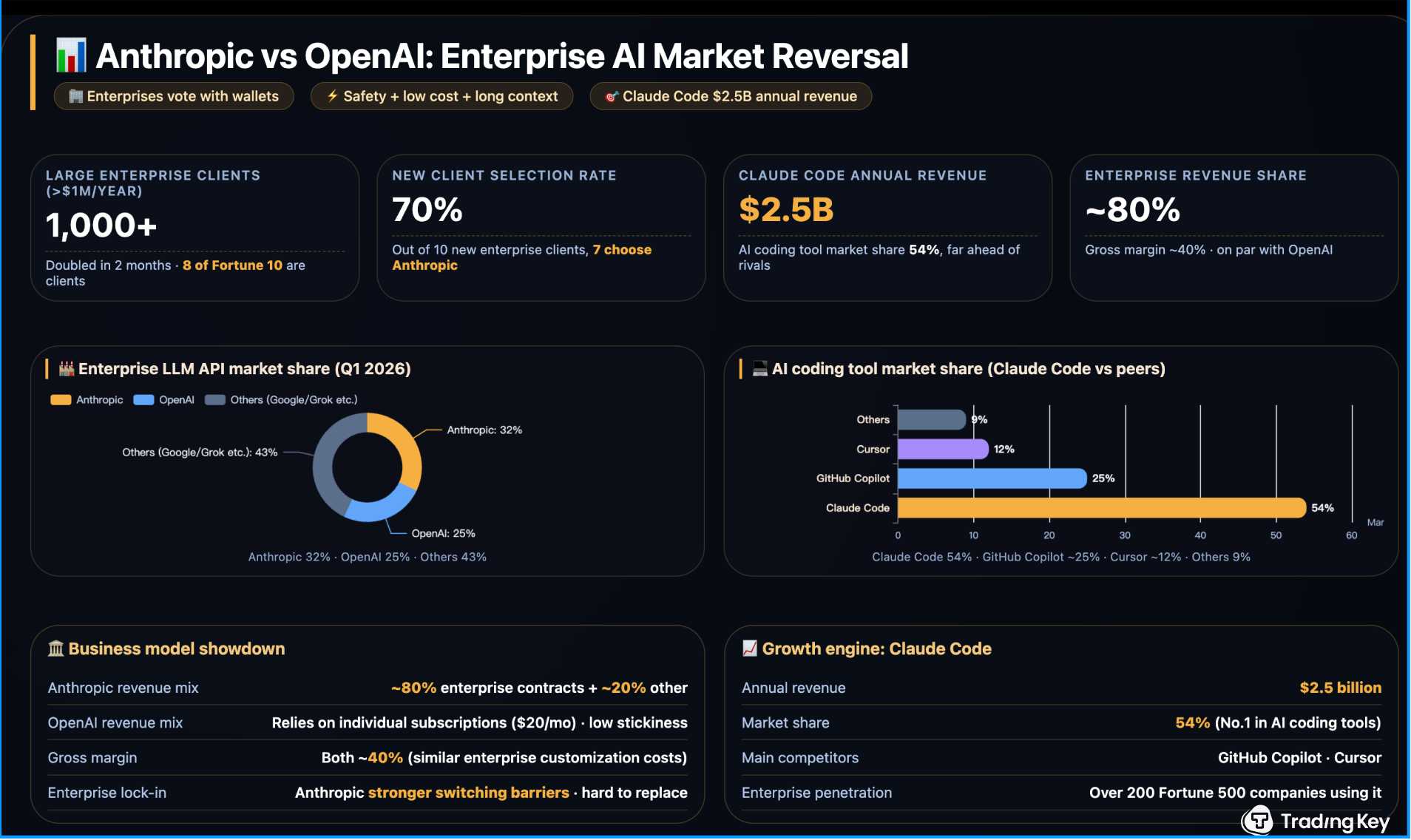
Anthropic Revenue Surpasses OpenAI for First Time, IPO as Early as October
Meta Is About to Launch a Consumer-Facing AI Model, and OpenAI Is About to Have Its IPO, Making Now a Good Time to Buy Meta Stock

Memory Chips Drive Samsung Earnings Surge, But Stock Price Opens High and Closes Lower: Why?

Is Bitcoin Reaching a Local Top? Institutional Demand vs. Retail Sentiment at $70,000

JPMorgan Warns Tesla Shares Could Fall Another 60%, Can Musk Still Turn the Tide?

Tradingkey







