ViroGates Submits De Novo Application To U.S. FDA For suPARnostic TurbiLatex
April 9 (Reuters) - ViroGates A/S VIROG.CO:
SAID ON WEDNESDAY SUBMITTED A DE NOVO MARKETING APPLICATION, FORMALLY REFERRED TO AS A "DE NOVO CLASSIFICATION REQUEST", TO THE U.S. FOOD AND DRUG ADMINISTRATION (FDA) FOR ITS SUPARNOSTIC TURBILATEX PRODUCT
RESTATES ITS PREVIOUSLY ANNOUNCED GUIDANCE AND EXPECTS 2026 REVENUE BETWEEN DKK 6 AND 7 MILLION AND AN EBIT OF DKK -10 TO -12 MILLION
Disclaimer: The information provided on this website is for educational and informational purposes only and should not be considered financial or investment advice.
Recommended Articles
Featured Tools
Top News
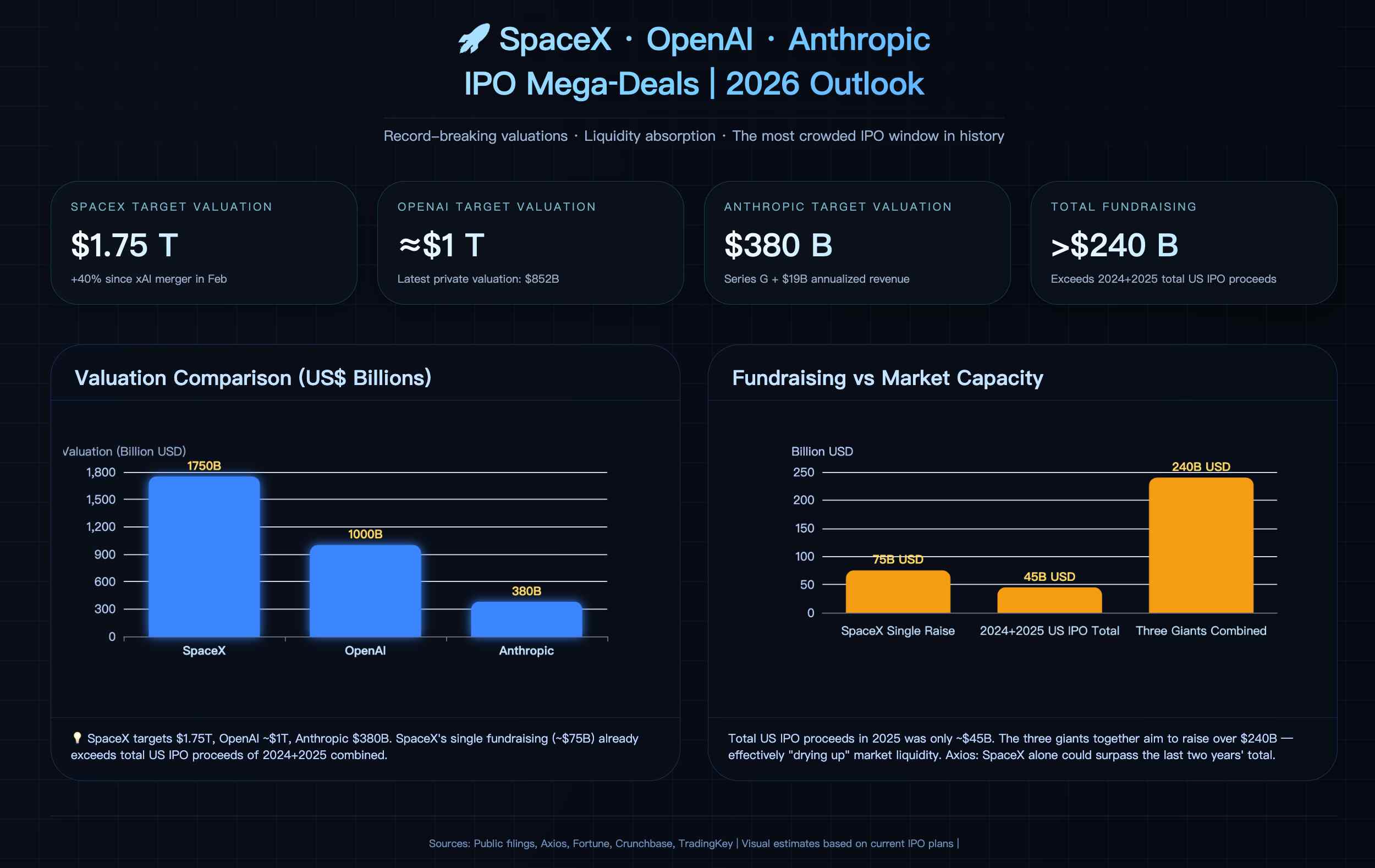
SpaceX IPO Guide: Can You Buy SpaceX IPO Shares on Robinhood? How Can Investors in Asia and Europe Buy SpaceX IPO Shares?

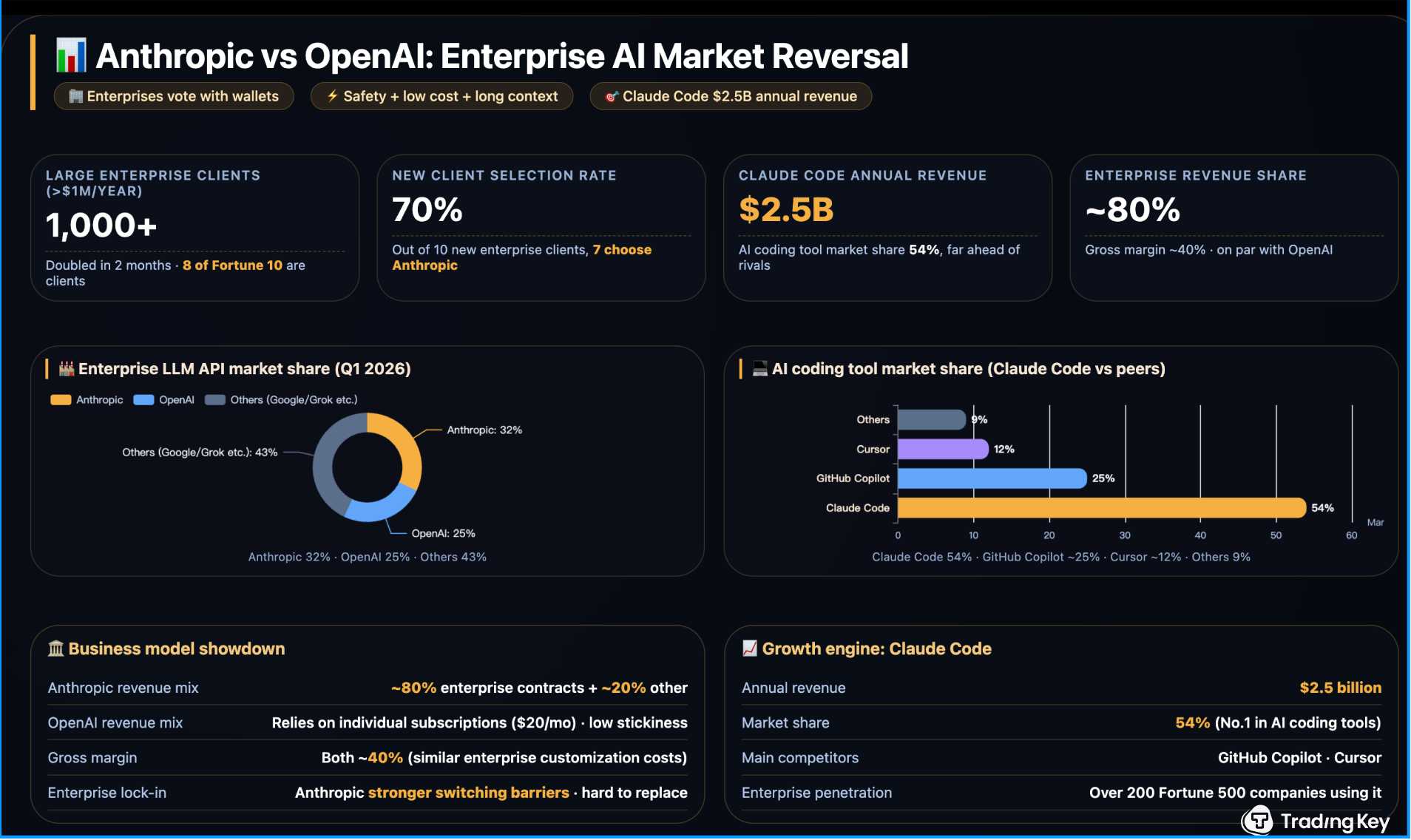
Anthropic Revenue Surpasses OpenAI for First Time, IPO as Early as October
Chevron Stock Analysis: A Stock Warren Buffett Bet Big On—Should You Invest in 2026 Despite High Oil Prices?

Meta Is About to Launch a Consumer-Facing AI Model, and OpenAI Is About to Have Its IPO, Making Now a Good Time to Buy Meta Stock

IonQ vs. Rigetti Computing: Which Quantum Computing Stock Has the Edge Now?

Tradingkey