Axsome Therapeutics Announces FOCUS Phase 3 Trial of Solriamfetol in Adults with Attention Deficit Hyperactivity Disorder (ADHD) Achieves Primary Endpoint
March 25 (Reuters) - Axsome Therapeutics Inc AXSM.O:
AXSOME THERAPEUTICS ANNOUNCES FOCUS PHASE 3 TRIAL OF SOLRIAMFETOL IN ADULTS WITH ATTENTION DEFICIT HYPERACTIVITY DISORDER (ADHD) ACHIEVES PRIMARY ENDPOINT
AXSOME THERAPEUTICS INC: WELL TOLERATED WITH SAFETY PROFILE GENERALLY CONSISTENT WITH PRIOR SOLRIAMFETOL TRIALS
AXSOME THERAPEUTICS INC: STATISTICALLY SIGNIFICANT REDUCTION IN OVERALL ADHD DISEASE SEVERITY AS MEASURED BY CGI-S SCORE COMPARED TO PLACEBO
AXSOME THERAPEUTICS : STATISTICALLY SIGNIFICANT IMPROVEMENT IN ADHD SYMPTOMS AS MEASURED BY AISRS TOTAL SCORE COMPARED TO PLACEBO
Disclaimer: The information provided on this website is for educational and informational purposes only and should not be considered financial or investment advice.
Recommended Articles
Featured Tools
Top News
Musk’s XChat Is Coming to App Store, Meta, Apple and Finance World Are Trembling

What is the CLARITY Act? 2026 Guide to US Crypto Regulation and Stablecoin Yield Rules

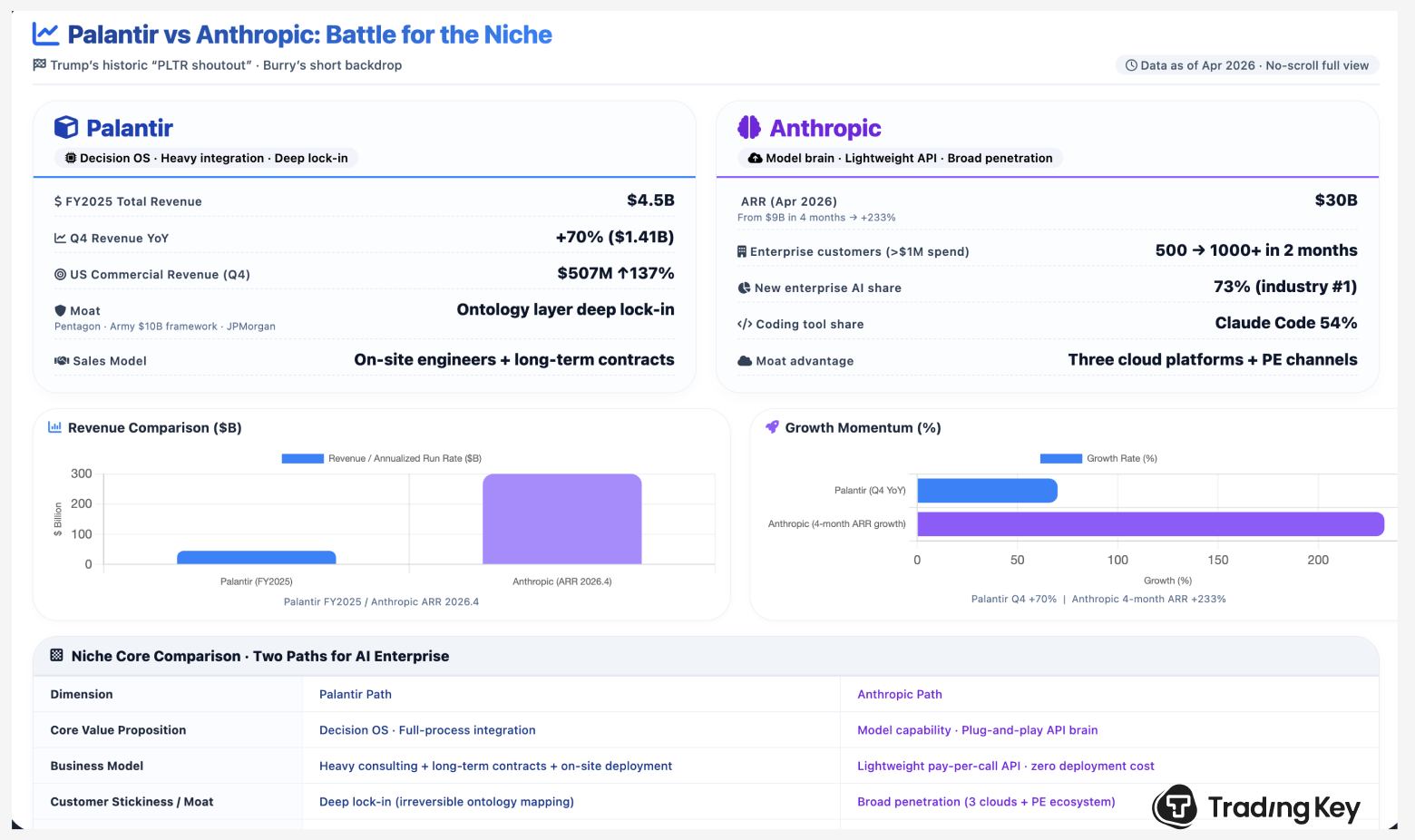
Anthropic IPO 2026: What the Claude Mythos Release Delay Means for Investors and Stock Valuation

Microsoft Launches Its Own AI Models and Ditches OpenAI Dependence - Is MSFT Stock a Buy at $370?

Anthropic Claude Mythos Preview Sparks Wall Street Panic: Bessent, Powell Summon CEOs; Cloudflare Tumbles 8%

Tradingkey