Axsome Therapeutics Inc Stock (AXSM) Moved Up by 10.20% on May 4: Facts Behind the Movement
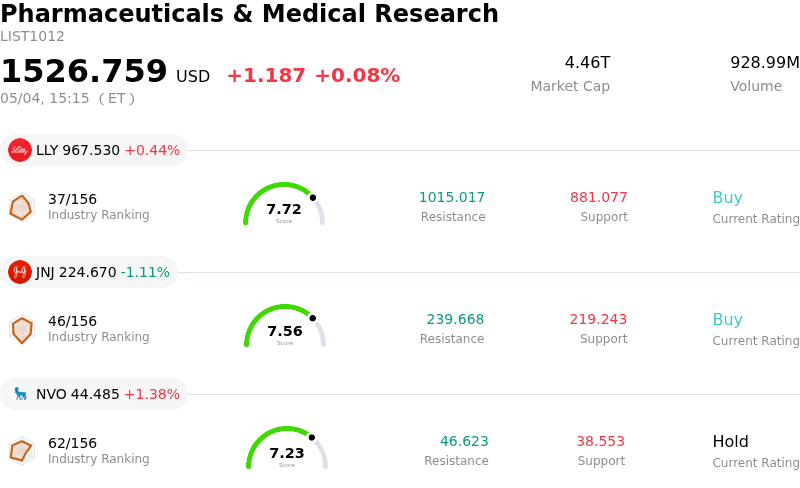
Axsome Therapeutics Inc (AXSM) moved up by 10.20%. The Pharmaceuticals & Medical Research sector is up by 0.08%. The company outperformed the industry. Top 3 stocks by turnover in the sector: Eli Lilly and Co (LLY) up 0.44%; Johnson & Johnson (JNJ) down 1.11%; Novo Nordisk A/S (NVO) up 1.38%.
What is driving Axsome Therapeutics Inc (AXSM)’s stock price up today?
Axsome Therapeutics (AXSM) experienced significant upward movement and intraday volatility, driven primarily by a combination of a major regulatory approval, robust financial performance, and positive analyst sentiment.
A key catalyst for the stock's performance was the U.S. Food and Drug Administration (FDA) approval of Auvelity for the treatment of agitation associated with dementia due to Alzheimer's disease. This approval occurred on April 30, 2026, marking a significant milestone as Auvelity is the first FDA-approved oral therapy specifically for this condition. The expanded indication for Auvelity significantly broadens its market potential, addressing a substantial patient population and offering a new treatment option where few previously existed. The FDA had granted Auvelity Breakthrough Therapy designation and Priority Review for this indication.
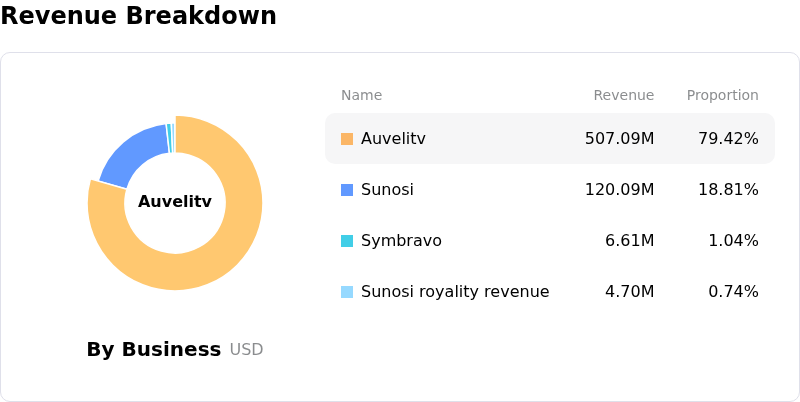
Furthermore, the company's release of its first quarter 2026 financial results on May 4, 2026, contributed to the positive sentiment. While Axsome Therapeutics reported a loss per share that was wider than analyst forecasts and revenue slightly below consensus, the market reacted favorably to strong year-over-year revenue growth. Total net product revenue for the quarter increased by 57% compared to the first quarter of 2025, with significant growth seen across its commercial products including Auvelity, Sunosi, and Symbravo. Investors appeared to prioritize the robust revenue growth and the strategic implications of the recent FDA approval over the short-term earnings per share miss.
Adding to the positive momentum, several financial analysts maintained or raised their price targets for AXSM around this period. For instance, HC Wainwright & Co. increased its price target for Axsome Therapeutics on May 4, 2026, maintaining a 'Buy' rating. Similarly, Needham had raised its price target for the company on May 1, 2026, also with a 'Buy' rating. The overall analyst consensus remains positive, with a majority recommending a 'Buy' rating, reflecting optimism about the company's future performance and growth potential in the biotechnology sector. The company also provided a business update highlighting other pipeline developments, such as the submission of a New Drug Application for AXS-12 and the addition of a new asset to its pipeline, further underpinning investor confidence in its long-term strategy.
Technical Analysis of Axsome Therapeutics Inc (AXSM)
Technically, Axsome Therapeutics Inc (AXSM) shows a MACD (12,26,9) value of [5.61], indicating a buy signal. The RSI at 73.21 suggests buy condition and the Williams %R at -27.08 suggests oversold condition. Please monitor closely.
Fundamental Analysis of Axsome Therapeutics Inc (AXSM)
Axsome Therapeutics Inc (AXSM) is in the Pharmaceuticals & Medical Research industry. Its latest annual revenue is $638.50M, ranking 59 in the industry. The net profit is $-183.17M, ranking 174 in the industry. Company Profile
Over the past month, multiple analysts have rated the company as Buy, with an average price target of $239.20, a high of $281.00, and a low of $200.00.
More details about Axsome Therapeutics Inc (AXSM)
Company Specific Risks:
- Axsome Therapeutics reported a Q1 2026 net loss of $1.26 per share, significantly missing analyst expectations of $0.86 per share, primarily due to a 53% surge in Selling, General, and Administrative (SG&A) expenses to $185.0 million, a level considered unsustainable long-term relative to revenue, and the company continues to operate with a negative operating margin and heavy leverage.
- Despite overall revenue growth, key marketed products Auvelity (for major depressive disorder) and Sunosi experienced sequential sales declines in Q1 2026, indicating potential challenges in maintaining or expanding market penetration for established therapies.
- The company faces significant commercial execution risk for the newly approved Auvelity indication for Alzheimer's agitation, as successful market penetration and real-world uptake are crucial to validate ambitious sales projections and could disappoint if launch metrics are not met.
Recommended Articles











