Regeneron Pharmaceuticals Inc Stock (REGN) Moved Down by 7.82% on Apr 29: Facts Behind the Movement
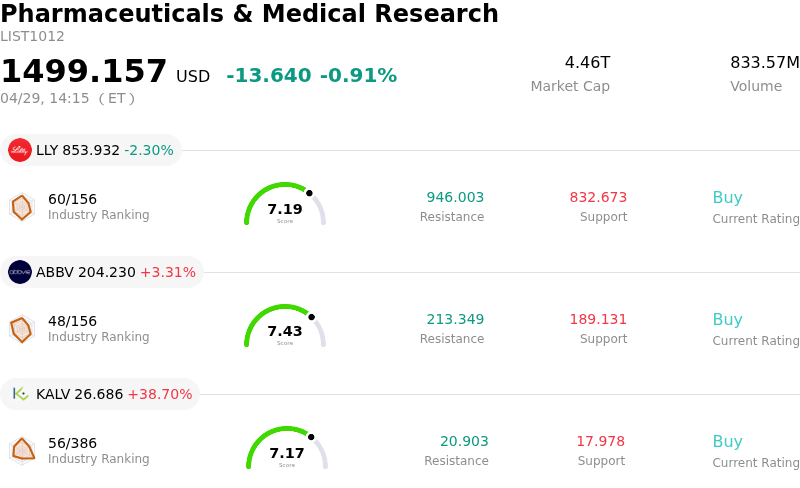
Regeneron Pharmaceuticals Inc (REGN) moved down by 7.82%. The Pharmaceuticals & Medical Research sector is down by 0.91%. The company underperformed the industry. Top 3 stocks by turnover in the sector: Eli Lilly and Co (LLY) down 2.30%; AbbVie Inc (ABBV) up 3.31%; Kalvista Pharmaceuticals Inc (KALV) up 38.70%.
What is driving Regeneron Pharmaceuticals Inc (REGN)’s stock price down today?
Regeneron Pharmaceuticals experienced a downward movement in its share price today, despite reporting a robust first quarter for 2026. The company announced strong financial results, with total revenues showing significant growth and non-GAAP earnings per share surpassing analyst expectations. This performance was largely driven by the continued strength of Dupixent global net sales and the approval of new indications for the drug. Additionally, Regeneron recently secured FDA approval for Otarmeni, a pioneering gene therapy for genetic hearing loss, and has committed to providing this therapy free of charge in the United States. The company also authorized a new substantial share repurchase program, signaling confidence in its future.
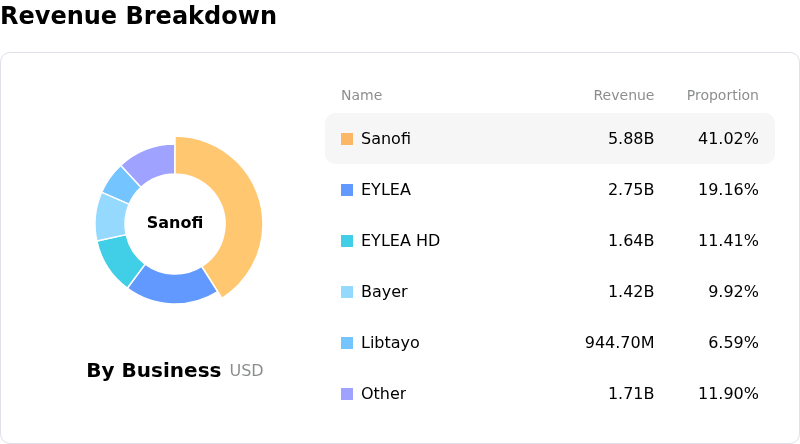
However, negative sentiment appears to have stemmed from several key factors, particularly related to the eye drug Eylea. Total U.S. net sales for Eylea and Eylea HD showed a decrease, with Eylea HD sales being negatively impacted by lower wholesaler inventory levels. More critically, regulatory complexities surrounding the Eylea HD pre-filled syringe weighed on investor confidence. The U.S. Food and Drug Administration did not act by its April target date on the company's application for a second contract manufacturer for the pre-filled syringe, leaving that application pending.
Furthermore, analyst commentary highlighted concerns regarding an experimental lung cancer drug combination that will not progress to late-stage trials. While the company achieved strong top-line growth, a decline in GAAP net income and a decrease in adjusted operating margin due to increased research and development, in-process research and development, and manufacturing costs also contributed to the cautious investor reaction. These elements collectively overshadowed the otherwise positive financial and pipeline developments, leading to the intraday price decline.
Technical Analysis of Regeneron Pharmaceuticals Inc (REGN)
Technically, Regeneron Pharmaceuticals Inc (REGN) shows a MACD (12,26,9) value of [-2.66], indicating a sell signal. The RSI at 40.75 suggests neutral condition and the Williams %R at -97.91 suggests oversold condition. Please monitor closely.
Fundamental Analysis of Regeneron Pharmaceuticals Inc (REGN)
Regeneron Pharmaceuticals Inc (REGN) is in the Pharmaceuticals & Medical Research industry. Its latest annual revenue is $14.34B, ranking 18 in the industry. The net profit is $4.50B, ranking 13 in the industry. Company Profile
Over the past month, multiple analysts have rated the company as Buy, with an average price target of $867.60, a high of $1057.00, and a low of $660.00.
More details about Regeneron Pharmaceuticals Inc (REGN)
Company Specific Risks:
- The U.S. Department of Justice "recently announced" a False Claims Act complaint, highlighted on April 22, 2026, alleging that Regeneron fraudulently inflated Medicare reimbursement rates for its key drug Eylea through undeclared price concessions between 2012 and 2021, posing a risk of substantial financial penalties and reputational harm.
- Regeneron's decision, announced on April 23, 2026, to provide its newly FDA-approved gene therapy, Otarmeni, for free in the U.S. introduces near-term revenue uncertainty for this innovative product, potentially impacting immediate financial projections despite the strategic intent.
- The ongoing DOJ False Claims Act complaint against Regeneron, concerning Eylea's Medicare pricing practices and recently highlighted on April 22, 2026, signals a potential for intensified regulatory scrutiny on drug pricing across the company's product portfolio.
Recommended Articles











