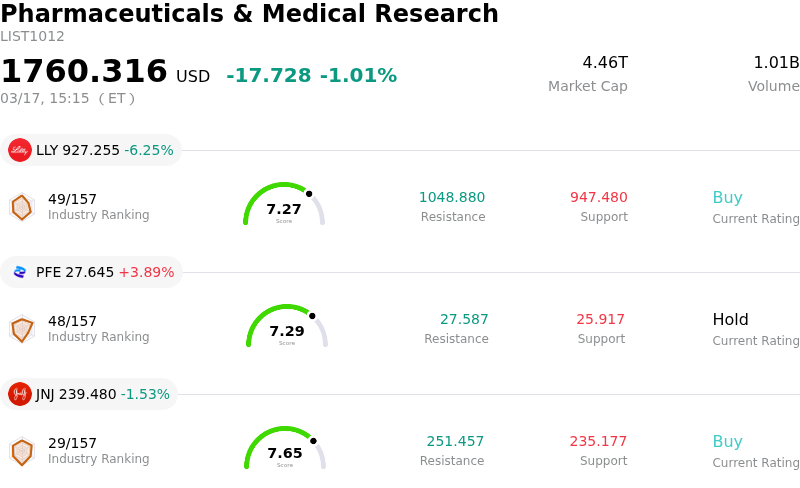
Pfizer Inc Stock (PFE) Moved Up by 3.89% on Mar 17: What Investors Need To Know
Pfizer Inc (PFE) moved up by 3.89%. The Pharmaceuticals & Medical Research sector is down by 1.01%. The company outperformed the industry. Top 3 stocks by turnover in the sector: Eli Lilly and Co (LLY) down 6.25%; Pfizer Inc (PFE) up 3.89%; Johnson & Johnson (JNJ) down 1.53%.
What is driving Pfizer Inc (PFE)’s stock price up today?
Pfizer Inc. (PFE) experienced an upward share price movement during intraday trading, influenced by several significant positive developments primarily related to its pipeline and analyst sentiment.
A key catalyst was the announcement of positive topline Phase 2 results for atirmociclib, Pfizer's next-generation CDK4 inhibitor for hormone receptor-positive, HER2-negative advanced or metastatic breast cancer. The FOURLIGHT-1 study met its primary endpoint, demonstrating a statistically significant and clinically meaningful improvement in progression-free survival, including a 40% reduction in the risk of disease progression or death. This investigational treatment also exhibited a manageable safety profile, reinforcing confidence in its potential, particularly as a successor to Pfizer's Ibrance.
Further positive news emerged from another Phase 2 study for tilrekimig, an investigational drug targeting moderate to severe atopic dermatitis, which also met its primary endpoint with significant improvements in patient symptoms. These clinical successes are crucial for Pfizer's oncology and immunology pipelines.
In response to these developments, analyst sentiment has shown a positive shift. HSBC maintained a Buy rating and raised its price target, indicating a positive outlook for the stock's future performance. BMO Capital also reiterated an Outperform rating for Pfizer following the positive tilrekimig data. Additionally, Argus Research upgraded Pfizer earlier in the month from a Hold to a Buy rating, signaling increasing confidence among some analysts.
Institutional investor activity further supported the upward trend, with firms like Focus Partners Wealth and Brighton Jones LLC significantly increasing their stakes in Pfizer during recent periods. The ongoing strategic focus on the obesity drug market, driven by the Metsera acquisition and its promising GLP-1 candidate, also continues to attract investor interest, anticipating a potentially disruptive monthly dosing option. These factors collectively contributed to the intraday price appreciation.
Technical Analysis of Pfizer Inc (PFE)
Technically, Pfizer Inc (PFE) shows a MACD (12,26,9) value of [0.14], indicating a neutral signal. The RSI at 47.02 suggests neutral condition and the Williams %R at -72.11 suggests oversold condition. Please monitor closely.
Fundamental Analysis of Pfizer Inc (PFE)
Pfizer Inc (PFE) is in the Pharmaceuticals & Medical Research industry. Its latest annual revenue is $62.58B, ranking 6 in the industry. The net profit is $7.77B, ranking 9 in the industry. Company Profile
Over the past month, multiple analysts have rated the company as Hold, with an average price target of $28.66, a high of $37.54, and a low of $24.00.
More details about Pfizer Inc (PFE)
Company Specific Risks:
- Analyst commentary from March 11, 2026, indicates a significant looming patent cliff, affecting approximately 25% of Pfizer's revenue and net income, which is expected to cause persistent earnings per share (EPS) volatility.
- Bearish analyst sentiment as of March 5, 2026, points to underwhelming progress in Pfizer's obesity drug pipeline and increased R&D intensity, leading to pressure on earnings and downward revisions of normalized EPS estimates.
- Concerns expressed by Pfizer's CEO, Alfred Bourla, on March 2, 2026 (reported March 9, 2026), regarding FDA decision-making and a specific official's conduct, suggest potential regulatory friction that could impact future product development and approvals.








