Tonix Pharmaceuticals surges after FDA nod for non-opioid pain drug
Drugmaker Tonix Pharmaceuticals' TNXP.O shares surge 19.5% to $60.83 in extended trading
U.S. Food and Drug Administration approves co's drug, Tonmya, to manage pain related to type of chronic condition called fibromyalgia
Fibromyalgia causes widespread pain, fatigue as well as problems with sleep and memory
Approval paves the way for the drug to be first new treatment option in over 15 years for patients
The pill is designed for bedtime treatment to improve sleep quality and reduce pain associated with the chronic condition
Stock up 55.7% YTD
Disclaimer: The information provided on this website is for educational and informational purposes only and should not be considered financial or investment advice.
Recommended Articles
Featured Tools
Top News
What is the CLARITY Act? 2026 Guide to US Crypto Regulation and Stablecoin Yield Rules

Why Meta Stock Jumped Today — And Whether 2026 Is Still a Time to Buy

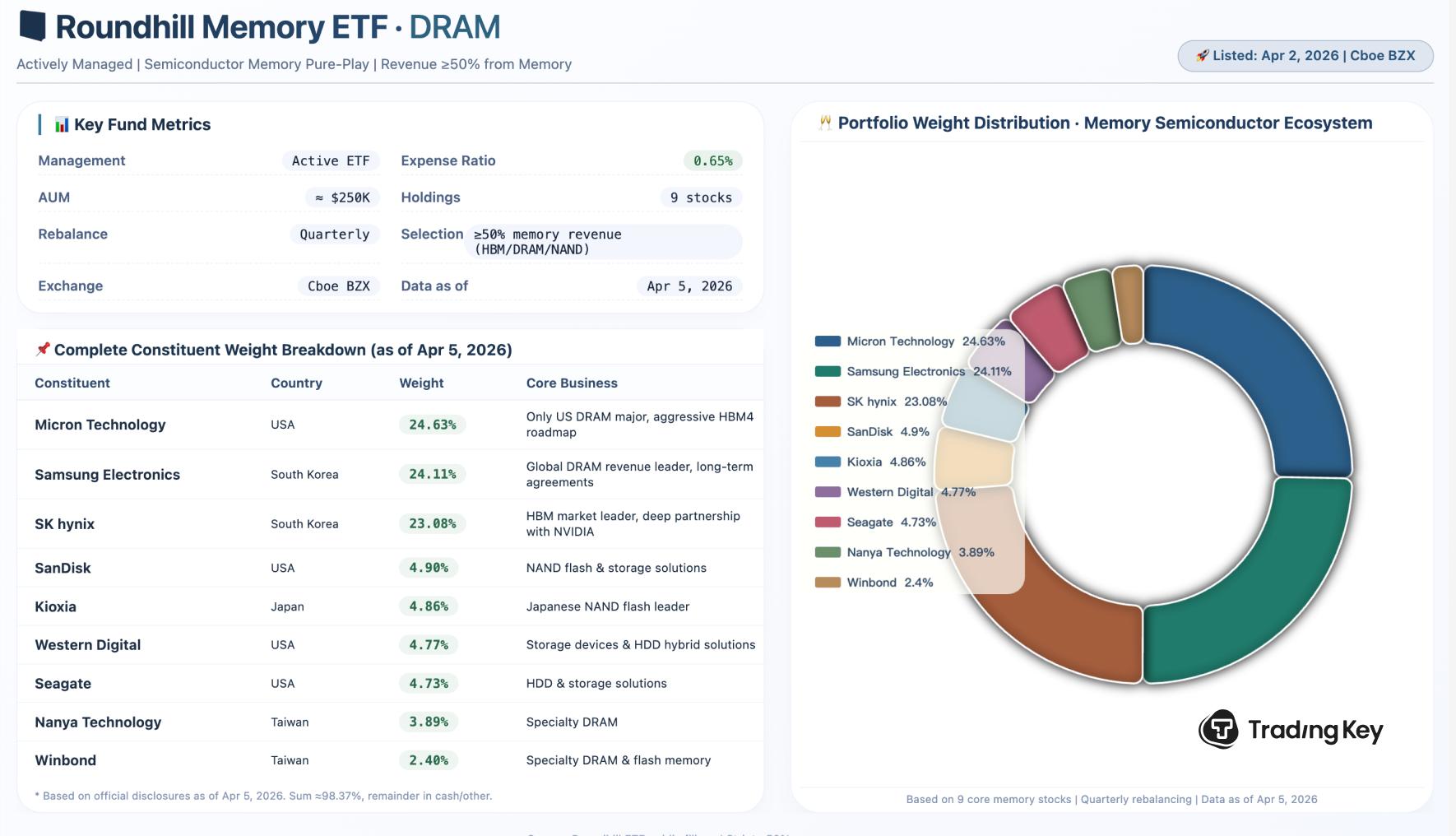
DRAM ETF Surges Over 18% in Overnight Trading, Is It Worth Investing?
Anthropic Claude Mythos Preview Sparks Wall Street Panic: Bessent, Powell Summon CEOs; Cloudflare Tumbles 8%

Anthropic IPO 2026: What the Claude Mythos Release Delay Means for Investors and Stock Valuation

Tradingkey







