Boston Scientific to not seek US approval for heart device amid regulatory challenges
May 28 (Reuters) - Boston Scientific BSX.N said on Wednesday it will stop pursuing U.S. approval for its experimental heart device due to increased clinical and regulatory requirements.
Disclaimer: The information provided on this website is for educational and informational purposes only and should not be considered financial or investment advice.
Recommended Articles
Featured Tools
Top News
What is the CLARITY Act? 2026 Guide to US Crypto Regulation and Stablecoin Yield Rules

Why Meta Stock Jumped Today — And Whether 2026 Is Still a Time to Buy

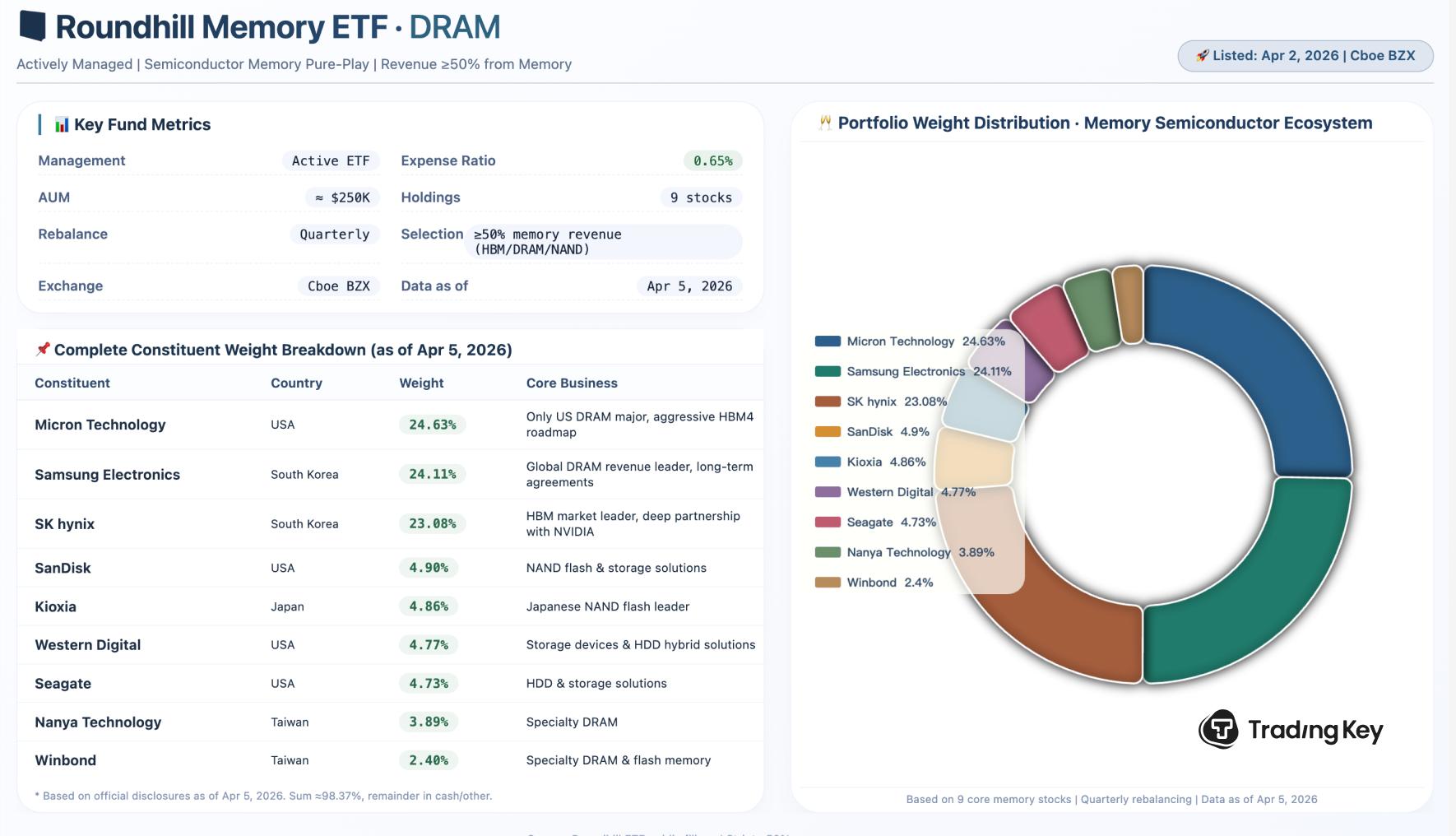
DRAM ETF Surges Over 18% in Overnight Trading, Is It Worth Investing?
Anthropic Claude Mythos Preview Sparks Wall Street Panic: Bessent, Powell Summon CEOs; Cloudflare Tumbles 8%

Anthropic IPO 2026: What the Claude Mythos Release Delay Means for Investors and Stock Valuation

Tradingkey







